Ocinaplon
- none
- pyridin-2-yl-(7-pyridin-4-ylpyrazolo[1,5-a]pyrimidin-3-yl)methanone
- 96604-21-6
 Y
Y
- 216456
- 4277
- 187602
 Y
Y
- 2H6KVC5E76
- D02617
 Y
Y
- DTXSID00242362

- Interactive image
- O=C(c1cnn2c(ccnc12)c3ccncc3)c4ncccc4
- InChI=1S/C17H11N5O/c23-16(14-3-1-2-7-19-14)13-11-21-22-15(6-10-20-17(13)22)12-4-8-18-9-5-12/h1-11H
 Y
Y - Key:OQJFBUOFGHPMSR-UHFFFAOYSA-N
 Y
Y
Ocinaplon is an anxiolytic drug in the pyrazolopyrimidine family of drugs. Other pyrazolopyrimidine drugs include zaleplon and indiplon.
Ocinaplon has a similar pharmacological profile to the benzodiazepine family of drugs, but with mainly anxiolytic properties and relatively little sedative or amnestic effect.[1]
Medical uses
A 2019 review found tentative evidence of benefit in anxiety.[2]
Mechanism of action
The mechanism of action by which ocinaplon produces its anxiolytic effects is by modulating GABAA receptors,[3] although ocinaplon is more subtype-selective than most benzodiazepines.[4]
Availability
Development of ocinaplon is discontinued due to liver complications that occurred in one of the Phase III subjects.[5]
Synthesis
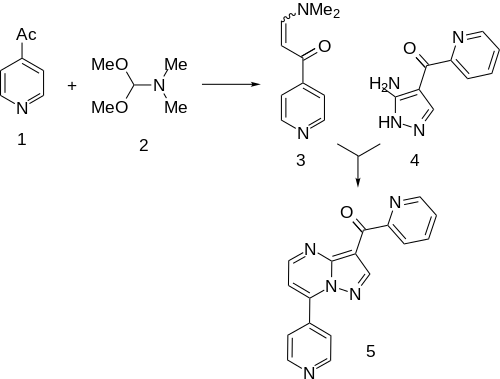
Condensation of 4-Acetylpyridine[8] with N,N-Dimethylformamide dimethyl acetal (DMFDMA) gives the "enamide" (3). This is then condensed with (3-Amino-1H-pyrazol-4-yl)(2-pyridinyl)methanone (4) (96219-90-8).[9][10] This is the same intermediate as was used in the synthesis of zaleplon in which the nitrile is replaced by a 2-acetylpyridil moiety. This affords the anxiolytic agent ocinaplon (5).
References
- ^ Lippa A, Czobor P, Stark J, Beer B, Kostakis E, Gravielle M, et al. (May 2005). "Selective anxiolysis produced by ocinaplon, a GABA(A) receptor modulator". Proceedings of the National Academy of Sciences of the United States of America. 102 (20): 7380–5. Bibcode:2005PNAS..102.7380L. doi:10.1073/pnas.0502579102. PMC 1129138. PMID 15870187.
- ^ Slee A, Nazareth I, Bondaronek P, Liu Y, Cheng Z, Freemantle N (February 2019). "Pharmacological treatments for generalised anxiety disorder: a systematic review and network meta-analysis". Lancet. 393 (10173): 768–777. doi:10.1016/S0140-6736(18)31793-8. PMID 30712879. S2CID 72332967.
- ^ Mirza NR, Rodgers RJ, Mathiasen LS (March 2006). "Comparative cue generalization profiles of L-838, 417, SL651498, zolpidem, CL218,872, ocinaplon, bretazenil, zopiclone, and various benzodiazepines in chlordiazepoxide and zolpidem drug discrimination". The Journal of Pharmacology and Experimental Therapeutics. 316 (3): 1291–9. doi:10.1124/jpet.105.094003. PMID 16339395. S2CID 21913400.
- ^ Atack JR (May 2005). "The benzodiazepine binding site of GABA(A) receptors as a target for the development of novel anxiolytics". Expert Opinion on Investigational Drugs. 14 (5): 601–18. doi:10.1517/13543784.14.5.601. PMID 15926867. S2CID 22793644.
- ^ "DOV Pharmaceutical, Inc. Places Ocinaplon Phase III Clinical Trial On Hold". PR NewsWire. Archived from the original on 4 March 2016.
- ^ Baumann M, Baxendale IR (October 2013). "An overview of the synthetic routes to the best selling drugs containing 6-membered heterocycles". Beilstein Journal of Organic Chemistry. 9: 2265–319. doi:10.3762/bjoc.9.265. PMC 3817479. PMID 24204439.
- ^ ARKIVOC 2010 (ii) 267-282
- ^ LaMattina JL, Sulesk RT (1986). "A-Amino Acetals: 2,2-Diethoxy-2-(4-Pyridyl)Ethylamine". Organic Syntheses. 64: 19. doi:10.15227/orgsyn.064.0019.
- ^ U.S. patent 4,900,836
- ^ CA 1243029
- v
- t
- e
- Benzodiazepines: Adinazolam
- Alprazolam
- Bromazepam
- Camazepam
- Chlordiazepoxide
- Clobazam
- Clonazepam
- Clorazepate
- Clotiazepam
- Cloxazolam
- Diazepam#
- Ethyl loflazepate
- Etizolam
- Fludiazepam
- Halazepam
- Ketazolam
- Lorazepam#
- Medazepam
- Nordazepam
- Oxazepam
- Pinazepam
- Prazepam; Others: Alpidem‡
- Barbiturates (e.g., phenobarbital)
- Carbamates (e.g., meprobamate)
- Carisoprodol
- Chlormezanone‡
- Ethanol (alcohol)
- Etifoxine
(α2δ VDCC blockers)
- SSRIsTooltip Selective serotonin reuptake inhibitors (e.g., escitalopram)
- SNRIsTooltip Serotonin-norepinephrine reuptake inhibitors (e.g., duloxetine)
- SARIsTooltip Serotonin antagonist and reuptake inhibitors (e.g., trazodone)
- TCAsTooltip Tricyclic antidepressants (e.g., clomipramine#)
- TeCAsTooltip Tetracyclic antidepressants (e.g., mirtazapine)
- MAOIsTooltip Monoamine oxidase inhibitors (e.g., phenelzine); Others: Agomelatine
- Bupropion
- Tianeptine
- Vilazodone
- Vortioxetine
(Antiadrenergics)
- Alpha-1 blockers (e.g., prazosin)
- Alpha-2 agonists (e.g., clonidine, dexmedetomidine, guanfacine)
- Beta blockers (e.g., propranolol)
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III











