BML-190
Chemical compound
 | |
| Identifiers | |
|---|---|
| |
| CAS Number |
|
| PubChem CID |
|
| ChemSpider |
|
| CompTox Dashboard (EPA) |
|
| Chemical and physical data | |
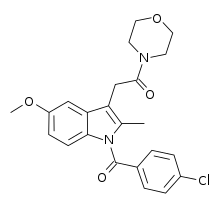
| Formula | C23H23ClN2O4 |
| Molar mass | 426.90 g·mol−1 |
| 3D model (JSmol) |
|
| |
InChI
| |
 N N Y (what is this?) (verify) Y (what is this?) (verify) | |
BML-190 (Indomethacin morpholinylamide) is a drug used in scientific research that acts as a selective CB2 inverse agonist.[1] BML-190 is structurally derived from the NSAID indomethacin but has a quite different biological activity.[2] The activity produced by this compound is disputed, with some sources referring to it as a CB2 agonist rather than an inverse agonist;[3][4] this may reflect an error in classification, or alternatively it may produce different effects in different tissues, and more research is required to resolve this dispute.
References
- ^ New DC, Wong YH (February 2003). "BML-190 and AM251 act as inverse agonists at the human cannabinoid CB2 receptor: signalling via cAMP and inositol phosphates". FEBS Letters. 536 (1–3): 157–60. doi:10.1016/S0014-5793(03)00048-6. PMID 12586356. S2CID 38569901.
- ^ Klegeris A, Bissonnette CJ, McGeer PL (June 2003). "Reduction of human monocytic cell neurotoxicity and cytokine secretion by ligands of the cannabinoid-type CB2 receptor". British Journal of Pharmacology. 139 (4): 775–86. doi:10.1038/sj.bjp.0705304. PMC 1573900. PMID 12813001.
- ^ Melck D, De Petrocellis L, Orlando P, Bisogno T, Laezza C, Bifulco M, Di Marzo V (January 2000). "Suppression of nerve growth factor Trk receptors and prolactin receptors by endocannabinoids leads to inhibition of human breast and prostate cancer cell proliferation". Endocrinology. 141 (1): 118–26. doi:10.1210/endo.141.1.7239. PMID 10614630.
- ^ Scutt A, Williamson EM (January 2007). "Cannabinoids stimulate fibroblastic colony formation by bone marrow cells indirectly via CB2 receptors". Calcified Tissue International. 80 (1): 50–9. doi:10.1007/s00223-006-0171-7. PMID 17205329. S2CID 23624771.
- v
- t
- e
Cannabinoids
(comparison)
| Cannabibutols |
|
|---|---|
| Cannabichromenes | |
| Cannabicyclols |
|
| Cannabidiols | |
| Cannabielsoins |
|
| Cannabigerols | |
| Cannabiphorols |
|
| Cannabinols | |
| Cannabitriols |
|
| Cannabivarins |
|
| Delta-8-tetrahydrocannabinols |
|
| Delta-9-tetrahydrocannabinols | |
| Delta-10-Tetrahydrocannabinols | |
| Miscellaneous cannabinoids |
|
| Active metabolites |
- Arachidonoyl ethanolamide (AEA; anandamide)
- 2-Arachidonoylglycerol (2-AG)
- 2-Arachidonyl glyceryl ether (2-AGE; noladin ether)
- 2-Oleoylglycerol (2-OG)
- N-Arachidonoyl dopamine (NADA)
- N-Arachidonylglycine (NAGly)
- 2-Arachidonoyl lysophosphatidylinositol (2-ALPI)
- N-Arachidonoyl serotonin (AA-5-HT)
- Docosatetraenoylethanolamide (DEA)
- Lysophosphatidylinositol (LPI)
- Oleamide
- Oleoylethanolamide (OEA)
- Palmitoylethanolamide (PEA)
- RVD-Hpα
- Stearoylethanolamide (SEA)
- O-Arachidonoyl ethanolamine (O-AEA; virodhamine)
cannabinoid
receptor
agonists /
neocannabinoids
| Classical cannabinoids (dibenzopyrans) |
|
|---|---|
| Non-classical cannabinoids |
|
| Adamantoylindoles |
|
| Benzimidazoles | |
| Benzoylindoles |
|
| Cyclohexylphenols | |
| Eicosanoids |
|
| Hydrocarbons | |
| Indazole carboxamides | |
| Indazole-3- carboxamides |
|
| Indole-3-carboxamides |
|
| Indole-3-carboxylates | |
| Naphthoylindazoles | |
| Naphthoylindoles |
|
| Naphthoylpyrroles | |
| Naphthylmethylindenes | |
| Naphthylmethylindoles | |
| Phenylacetylindoles | |
| Pyrazolecarboxamides |
|
| Pyrrolobenzoxazines | |
| Quinolinyl esters | |
| Tetramethylcyclo- propanoylindazoles | |
| Tetramethylcyclo- propanoylindoles | |
| Tetramethylcyclo- propylindoles | |
| Others |
|
enhancers
(inactivation inhibitors)
- 4-Nonylphenylboronic acid
- AM-404
- Arachidonoyl serotonin
- Arvanil
- BIA 10-2474
- Biochanin A
- CAY-10401
- CAY-10429
- Genistein
- Guineesine
- IDFP
- JNJ 1661010
- JNJ-42165279
- JZL184
- JZL195
- Kaempferol
- LY-2183240
- MK-4409
- O-1624
- O-2093
- Oleoylethanolamide (OEA)
- Olvanil
- Palmitoylethanolamide (PEA)
- PF-04457845
- PF-622
- PF-750
- PF-3845
- PHOP
- URB-447
- URB-597
- URB-602
- URB-754
- VDM-11
(antagonists/inverse
agonists/antibodies)
- AM-251
- AM-281
- AM-630
- AM-1387
- AM-4113
- AM-6527
- AM-6545
- BML-190
- Brizantin (Бризантин)
- CAY-10508
- CB-25
- CB-52
- CB-86
- Dietressa (Диетресса)
- Drinabant (AVE1625)
- Hemopressin
- Ibipinabant (SLV319)
- JTE-907
- LH-21
- LY-320,135
- MDA-77
- MJ-15
- MK-9470
- NESS-0327
- NIDA-41020
- O-606
- O-1184
- O-1248
- O-1918
- O-2050
- O-2654
- Otenabant (CP-945,598)
- PF-514273
- PipISB
- PSB-SB-487
- Rimonabant (SR141716)
- Rosonabant (E-6776)
- SR-144,528
- Surinabant (SR147778)
- Taranabant (MK-0364)
- TM-38837
- VCHSR
- See also: Cannabinoid receptor modulators (cannabinoids by pharmacology)
- List of: AM cannabinoids
- JWH cannabinoids
- Designer drugs § Synthetic cannabimimetics
 | This cannabinoid related article is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e











